ANANDA Scientific Announces FDA approval of the IND for the Clinical Trial on the Treatment of Opioid Use Disorder (OUD)
Por um escritor misterioso
Descrição
ANANDA Scientific Inc., (a biotech pharma company) today announced approval by the U.S. Food and Drug Administration (FDA) of the Investigational New

Nantheia ATL5: A New Hope for Beating Opioid Addiction - Treatment Magazine

Sohail R. Zaidi posted on LinkedIn

ANANDA Scientific Announces FDA Approval of the IND for a Clinical Trial exploring treatment of Social Anxiety Disorder (SAD)

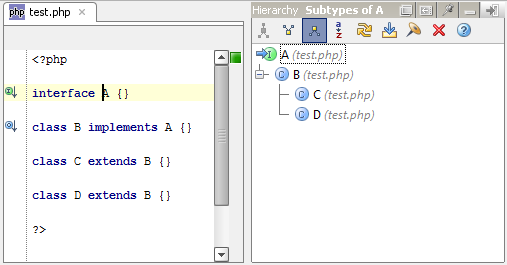
ANANDA Scientific
4 Medications for Opioid Use Disorder in Various Treatment Settings, Medications for Opioid Use Disorder Save Lives
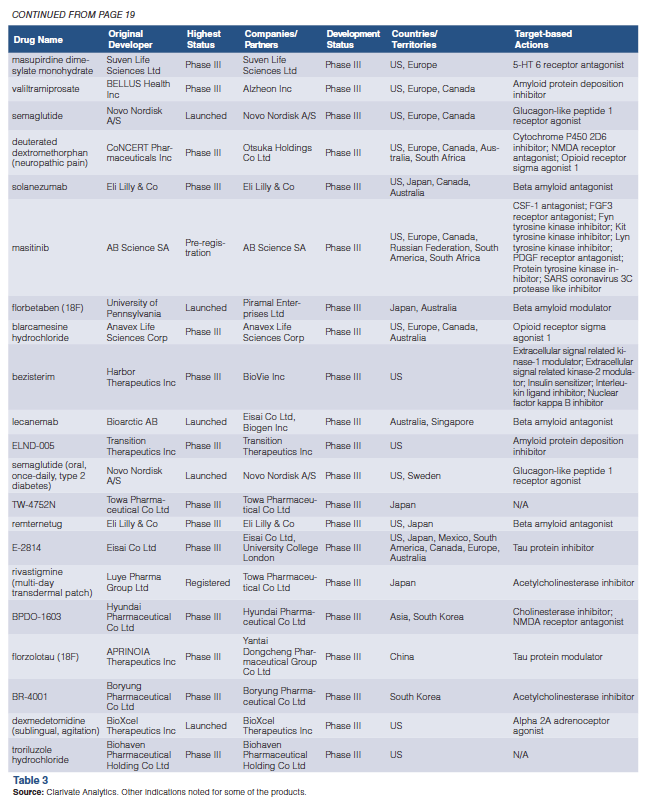
2024 Pipeline Report: First-World Focus

FDA approves IND for Ananda Scientific's PTSD treatment

ANANDA Scientific announces FDA approval of the IND for the clinical trial on the treatment of Opioid Use Disorder (OUD) - ANTARA News

A Decade of FDA-Approved Drugs (2010–2019): Trends and Future Directions
de
por adulto (o preço varia de acordo com o tamanho do grupo)