Designing an EDC System to Work for a CRA
Por um escritor misterioso
Descrição

Medrio, Inc.

Designing an EDC System to Work for a CRA
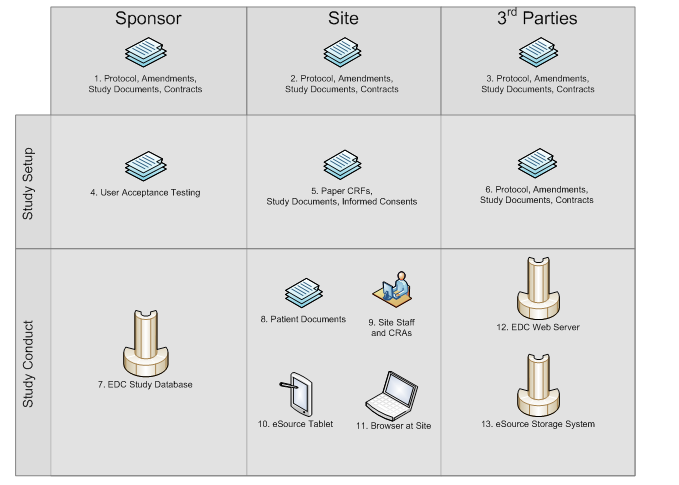
eSource Records in Clinical Research

Consider EDC For Designing and building Clinical Trials

Selecting the Right EDC System for Your Clinical Research

CRA (Clinical Research Associate)
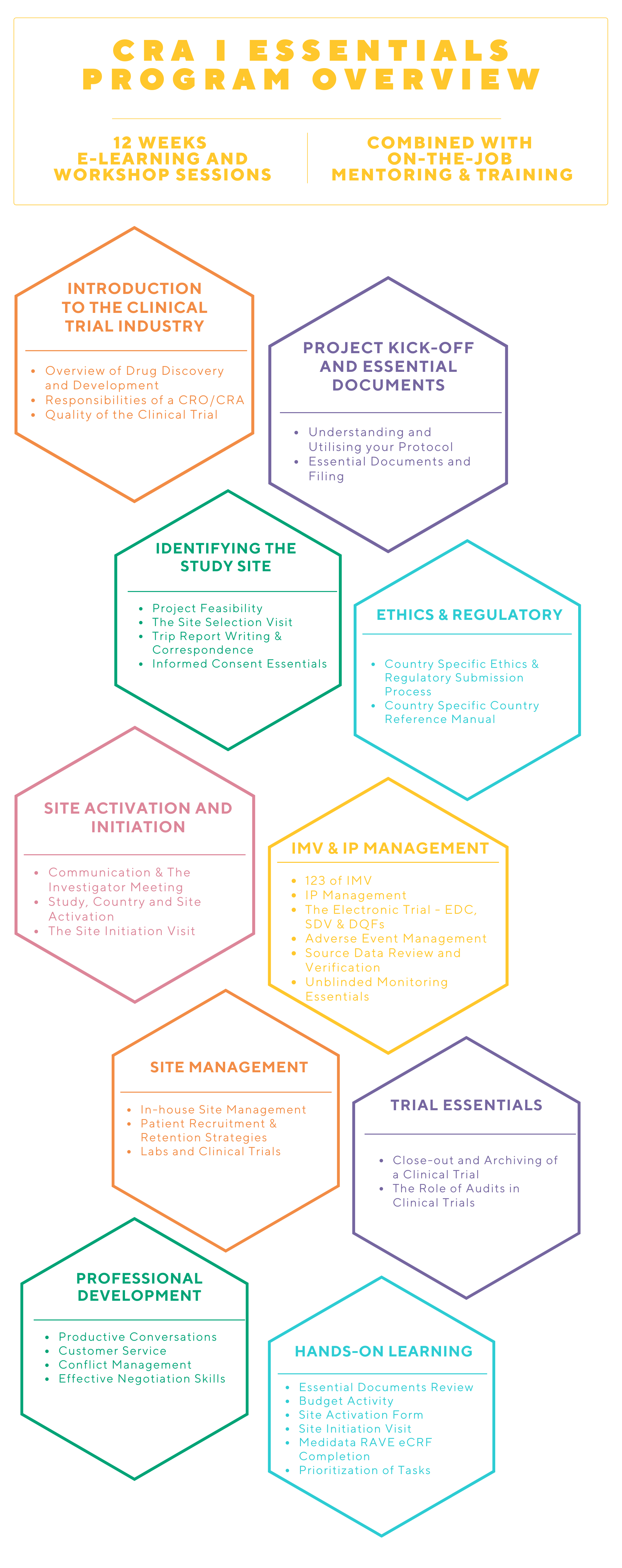
Clinical Research Associate (CRA) Essentials Program
ADVANCED CLINICAL TRIALS Services - ADVANCED CLINICAL TRIALS

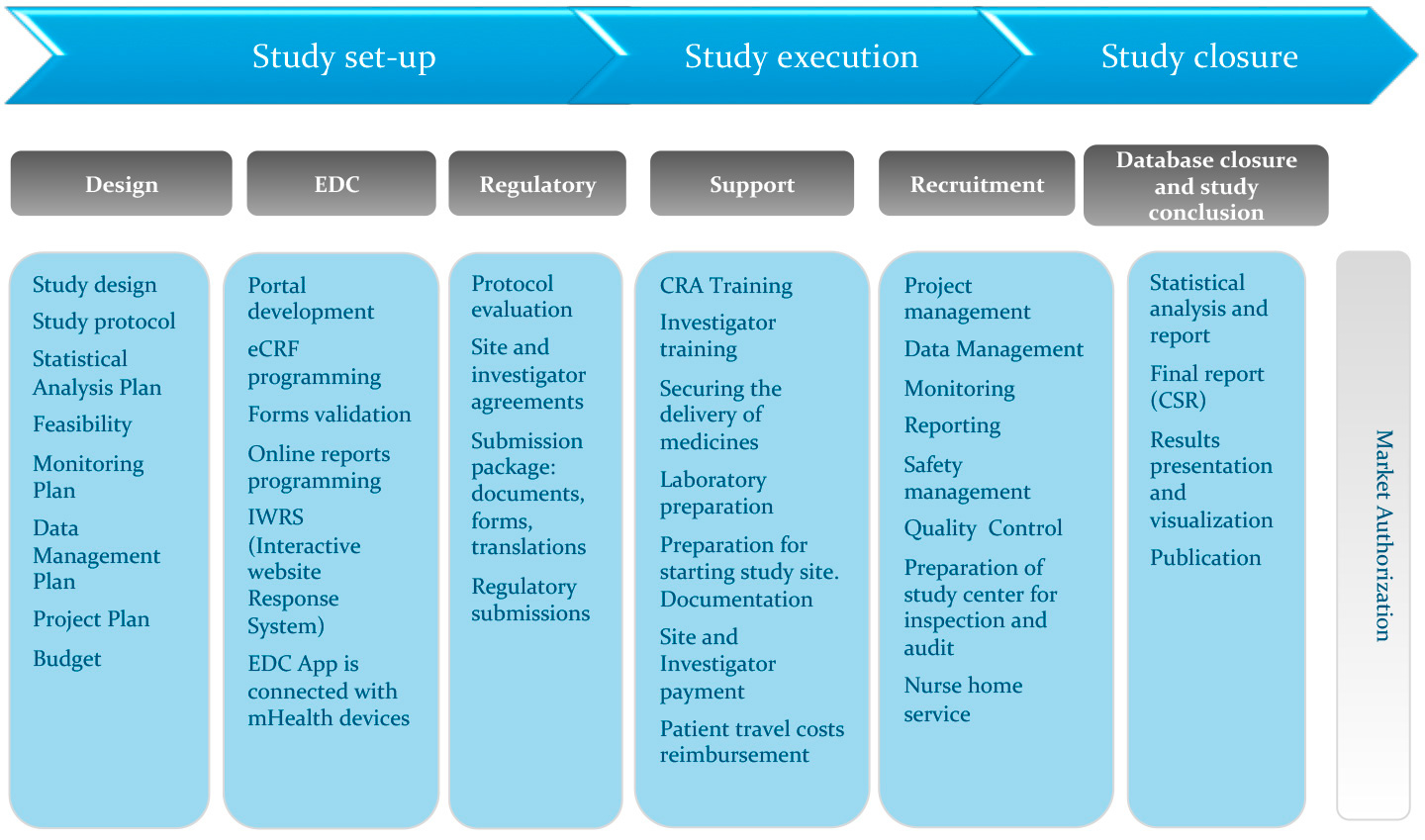
CTMS eTMF and EDC for Clinical Investigations

The CRA's Guide to Monitoring Clinical Research, Fifth Edition
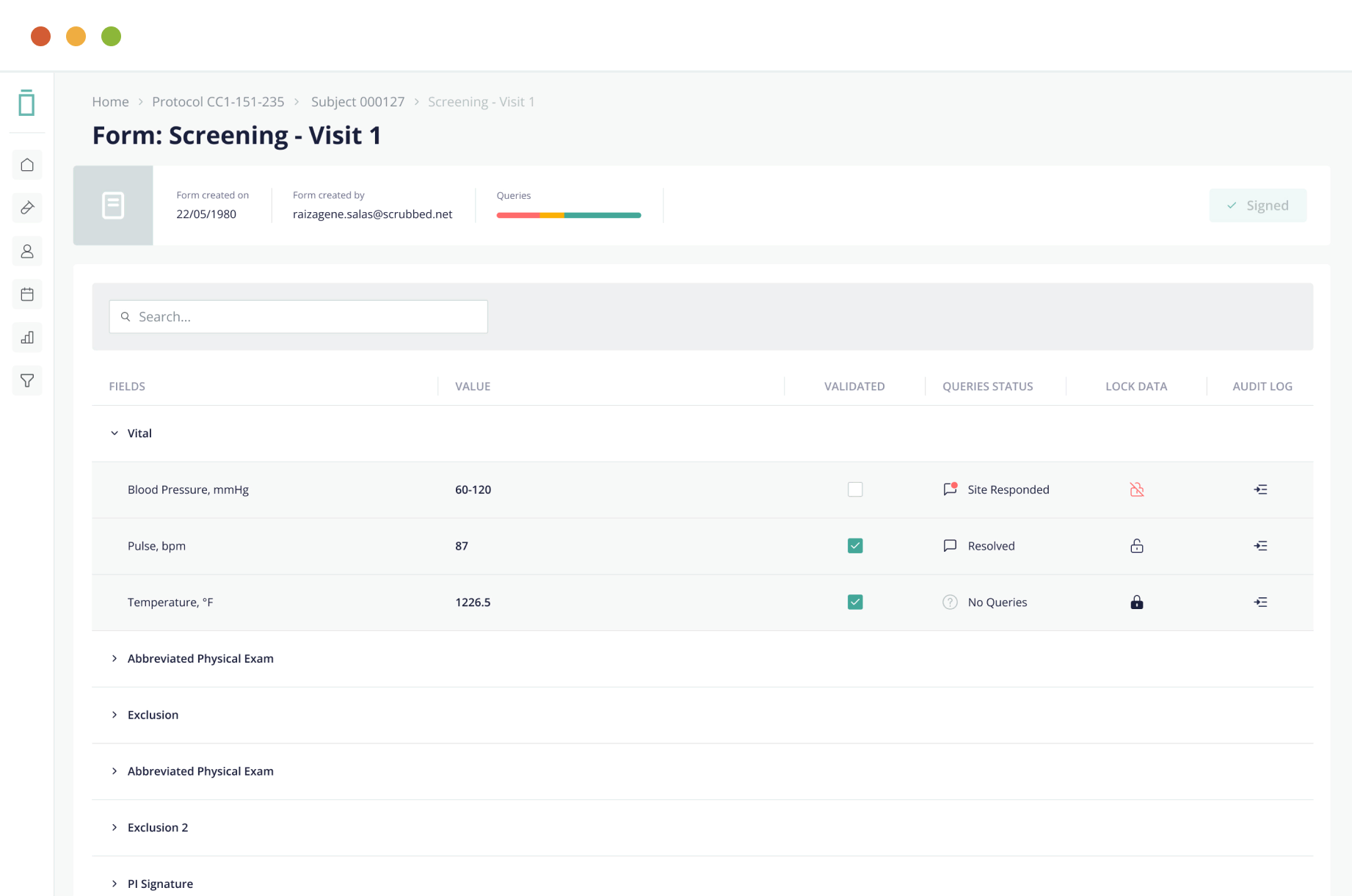
Electronic Source (eSource)

Clinical Research Associate Resume Sample

Cyrus Theuri, RN, MPH, CCRP on LinkedIn: #school #share #canada
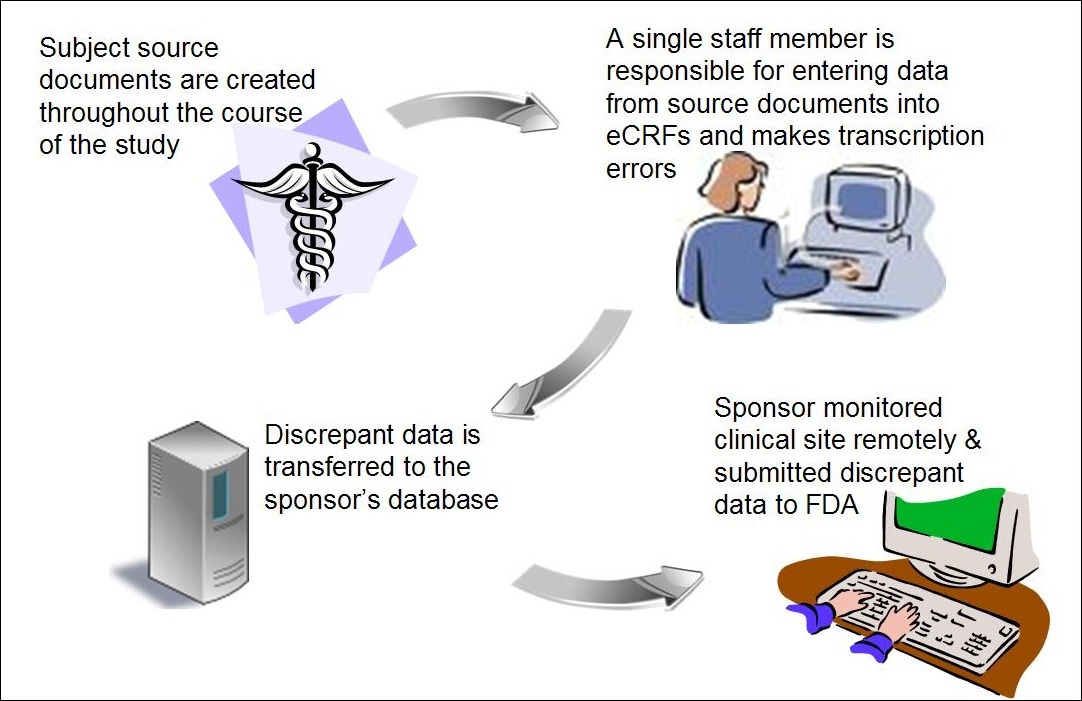
The Future of Clinical Trials Using Electronic Data Capture
de
por adulto (o preço varia de acordo com o tamanho do grupo)